Compound Interest, http://www.compoundchem.com/2013/12/30/the-chemistry-of-fireworks/
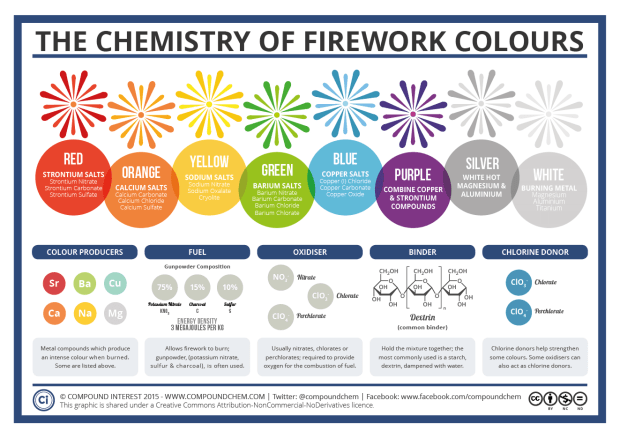
The 4th of July fireworks that we saw last night would not be possible without minerals. Fireworks mainly contain gunpowder, which is a combination of charcoal, sulfur, and the mineral potassium nitrate. In order to create the pretty colors we are used to seeing in fireworks, mineral salts are added. This infographic from Compound Interest explains which mineral salts create which colors. If you go to the website, you can read a lot more about the chemistry of fireworks and a brief explanation of why different minerals make different colored flames.
I learned that blue fireworks are very difficult to produce because copper chloride breaks down at high temperatures, so they have to somehow keep the temperature hot enough to ignite but not so hot that the blue color vanishes. Thus, you almost never see purple fireworks because it is a combination of red and blue.












