




Posted by April Buettner – member of Show-Me Rockhounds of Kansas City
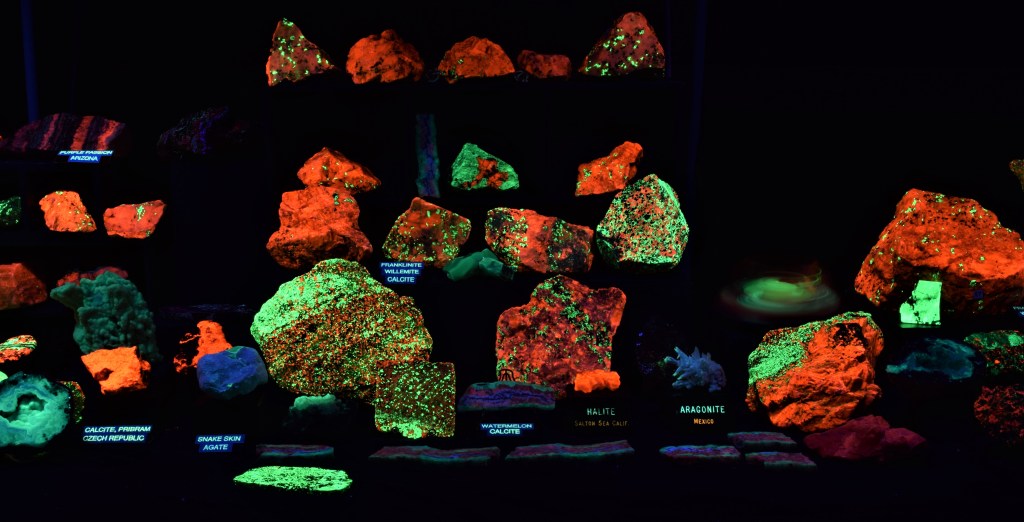
The Show-Me Rockhounds went to Baldwin City, Kansas and bought some rocks and minerals from an old time rockhound. Here are some fluorescent minerals that I bought. Photographed under short-wave UV light and white light.


From Left to Right:
Back row: Uranyl activated Quartz; Three Purple Passion minerals from the Purple Passion Mine in Arizona. Orange is Calcite, blue is Fluorite and green is Willemite.
Middle Row: Granite with Mica; Purple Passion minerals;
Front Row: Agate; Chalcedony; Unknown “Bacon” Rock
From the collection of and photographed by April Buettner
HERE IS A VARIETY OF FLUORESCENT MINERALS THAT WILL BE AT THE 61ST GEM AND MINERAL SHOW. SPONSORED BY THE ASSOCIATION OF EARTH SCIENCE CLUBS OF KANSAS CITY. MARCH 10,11,12, 2023 AT THE KCI EXPO CENTER, 11730 AMBASSADOR DRIVE, KANSAS CITY, MO. DISPLAYED BY DAN AND CONNIE SNOW OF THE SHOW-ME ROCKHOUNDS OF KANSAS CITY.







Introduction to the Mary Bergman Hamilton Collection of Minerals
In 2017, the children of Mary Bergman Hamilton donated her lifetime collection of minerals to the Show-Me Rockhounds. In order to share this gift with other rockhounds and interested earth science students, our club has put together this publication.
Mary had divided her collection into minerals she had purchased, those she had traded for, and those that she had found on rockhounding field trips. We have followed this same format in how we have organized this book. At the beginning of each section we have some tips for how you, as a rockhound, can add to your collection using these methods.
A number of the minerals in the collection fluoresce under short- or long-wave ultraviolet light. We have added a section to the book to highlight these minerals and explain a little about fluorescence.
We have a page showing the collection as it was displayed at the 60th Annual Kansas City Gem and Mineral Show in 2022.
Finally, there is a page of the book to emphasize the importance of good recordkeeping and labeling of a mineral collection. We show Mary’s method of index cards and mineral labeling.
We hope that this book will inspire both novice and experienced rockhounds by showing what a dedicated amateur can accomplish.

We have books available for $15.00 per copy. If you are interested in purchasing a copy(s) you may email Dan Snow President of the Show-Me Rockhounds of Kansas City at showmerockhounds.com
ALL PHOTOS BY Dan or Connie Snow










PHOTOS BY DAN SNOW



These are pictures of a few selections in our collection. The Scolecite, Stilbite-Ca and Calcite (about 12 inches long) was from Sami Makki, whose grandfather started the rudiments of Matrix India in the 50’s. Sami’s father Muhammad and he now run the operation that began exporting minerals in the 70’s. Sami personally collects much of the material and he personally collected this and other specimens we have. The specimen comes from the basalt plains in Maharashtra, India.
Sami was called by a construction crew about a potential pocket of minerals found during the digging of a well. (Sami and his team have networked with construction companies all over India and often get calls in addition to the mines they operate.) The well wall started gushing water and they quit for the day. Next day they realized they hit a pocket of water and called Matrix India to check it out before continuing to the actual water table. Sami said these Scolecite formations take millions of years of still water to form in. They then bring their own team in to remove the specimen material and reimburse the construction company.
I recently saw an article about research being done on these kinds of trapped water for bacteria that have been isolated for millions of years! The Vandinite is almost always found in oxidation zones around lead deposits (per Mindat.org). These come from Morocco and exhibit brown and reddish-brown, two of the many colors this mineral can have.


