
Images
November Show Coming Soon
 Only 2 weeks until the Fall Kansas City Gem Show! Here’s a flyer in PDF format that you can print and hang up wherever you like, and don’t forget to pass around your coupons, too.
Only 2 weeks until the Fall Kansas City Gem Show! Here’s a flyer in PDF format that you can print and hang up wherever you like, and don’t forget to pass around your coupons, too.
Chemistry in Mining
During Earth Science WeekTM, we went to a lecture by Dr. Innocent Pumure from UCM called “Sonochemical Extraction of Arsenic and Selenium from Pulverized Rocks Associated with Mountaintop Removal Valley Fill (MTR/VF) Method of Coal Mining”.
You may be wondering, what is Mountaintop Removal Valley Fill Mining? First, the excavation company blows up (or strips) the top part of the mountain to remove vegetation and expose the coal seams. The coal seams are then mined through the open cast/strip method, and the extra rock and soil is dumped in nearby valleys called valley fills. It is cheaper and easier to do than regular mining, where they dig a vertical shaft down and do everything through the tunnel, but it blasts the mountain apart and looks ugly. Since 30% of electricity in the USA comes from coal, valley fill mining is still pretty popular.
In 2002, the EPA found too much selenium downstream of a certain mine in West Virginia (we’re not going to say which one). It was over 5 ng/mL, which was the limit back then.[*] 7 years later, there was still an active mine there and the water still had too much selenium. Even worse, the surrounding sediment had 10.7 mg/kg selenium. This could cause problems for the environment later. Due to bioaccumulation, you could say once it’s in there, it’s really in there.
So now we get to the topic of Dr. Pumure’s talk, in which he and his colleagues discovered a way to quickly find out how much selenium and arsenic were in the ground around this mine in West Virginia. When you do a chemical analysis, you usually have to break down the samples in order to measure what is in them. One method to do this would be to take some core samples and do an acid extraction, but that takes a long time and uses a lot of reagents. Sonochemical extraction uses ultrasound energy to accelerate the leaching process that would naturally happen as rocks become weathered. Since it is ultrasound, it does not directly touch the sample, is minimally invasive, and does not need any reagents except water.
Next, he explained the methodology, which means a description of exactly how they did it in the lab: the size of the extraction cells, how much water and power were used (200W/cm3), how long the samples were sonicated, and all the other pertinent information for chemists. Pumure actually spent quite a lot of time finding out the optimal sonicating time to get the best extraction. It turned out the best times for his sample sizes were 20 minutes for Se and 25 minutes for As. That’s really fast![**] Then, he did a comparison to a chemical sequential extraction to make sure that the sonochemical extraction method was getting everything. To summarize, yes it was. Finally, he did a principal component analysis of core samples from different places all over the mountain using this same technique. They found some really interesting trends and correlations, for example, it appears that there is more arsenic in illite clay than other types of clay.
This research has many useful applications. If you were running a mine, you could take samples more frequently to see if your mine is polluting the surrounding environment, and then you could do something about it before the EPA finds out. The method could probably be used for other analytes, too. For other research needs, you could now quickly analyze large batches of mineral samples to get lots of data that would otherwise be too expensive or time consuming to obtain.
[*]The EPA has since lowered the limit and now it is 3.1 ng/mL.
[**]For comparison, some of my colleagues do chemical extractions that take 2 days.
Lesedi La Rona Diamond Sells

Photo by Donald Bowers/Getty Images for Sotheby’s
In the fall of 2015, a 1,109 carat white diamond was found in the Lucara mine in South Africa. The diamond is called the Lesedi La Rona, which means “Our Light” in Setswana. They tried to sell it at a Sotheby’s auction last year (July 2016), and we even wrote about it on Show Me Rockhounds, but no bidder met the reserve. Now, it has finally sold.
On September 26, 2017, Graff Diamonds announced that they bought the Lesedi La Rona for $53 million in a private sale. The CEO of Lucara says $53 million is higher than the highest bid they got at the auction last year, but he wishes he could have got a higher price. (Don’t we all!)
What will they do with it? Lawrence Graff, the founder of Graff Diamonds, says, “The stone will tell us its story, it will dictate how it wants to be cut, and we will take the utmost care to respect its exceptional properties. … I am privileged to be given the opportunity to honor the magnificent natural beauty of the Lesedi La Rona.”
https://www.cnbc.com/2017/09/26/large-diamond-the-lesedi-la-rona-sells-for-53-million.html
Scorodite

Photo by Yaiba Sakaguchi
This scorodite specimen is only 8mm long, but it is very pretty. It is from the Drakelands Mine, Plympton, Tavistock District, Devon, England, UK. It gets its name from the Greek σκορόδιου = “Scorodion”, which means it smells like garlic when heated. Scorodite can also be called Arsenic Sinter. It comes from the oxidation of arsenopyrite or other arsenic-bearing species.
Chemical Formula: Fe3+AsO4·2H2O
Association Picnic Photos 2017

Shelter House 3, although many people had already left. Photo by Stephanie Reed

Jim Ray, Stephanie, Martin, Valerie, and Jeanna. Photo by David Reed

Looking at something shiny. Photo by Stephanie Reed
Despite the rain, there was a sizeable turnout at the picnic, with members from Show-Me, IGAMS, the Bead Society, and more. Bruce, Martin, and Jim Ray grilled, with plenty of umbrella helpers keeping the rain off. Every picnic table in Shelter #3 was covered with interesting items during swap time, but it was cleared off quickly to make room for food and auction action. There was a lot of good food and if anyone went away hungry they have only themselves to blame. Kara was the auctioneer and sold things like trilobites, ammonites, calcite crystals, Dr. Gentile’s book (he was at the picnic, so the winner got it signed!), a specimen of garnets on chlorite schist, necklaces, beads, and even a set of all-beef hot dogs with matching buns. The proceeds will go towards the Scholarship Fund, which will be awarded next March.
Mini Field Trip July 2017
After our meeting on July 15, 2017 we decided to go on another mini field trip and look for fossils. There were crinoid stems, composita, and other fossils, as well as lots of blue shale. There was also something red and nobody knew what it was. I don’t have many pictures because it was very hot outside and my phone said something about battery temperature too high.

Composita found in Kansas City. Photo by Stephanie Reed

Blue shale. Photo by Stephanie Reed

David met a friendly bird. Photo by David Reed.

This is a traditional pose for members of rock clubs. Someday we may make a calendar. Photo by Stephanie Reed
Extremely Hard Water

Steve Hardinger and Dragon Minerals. Retrieved from Mindat Photo of the Day
Everyone knows Kansas City’s water tastes really good, despite being “hard water.” Kansas City municipal water averages 100 ppm (CaCO3 equivalent), which is classified as “moderately hard” by the Water Quality Association. But these people’s hard water would be off the charts!
A valve was stuck in a pipe somewhere in the Naica mining complex in Chihuahua, Mexico. “The valve was removed with the aid of a saw, and voila! the pipe was completely clogged with fine, colorless and gemmy gypsum crystals to 7 cm.”
No word on what they did with the crystals after they unclogged the pipe.
1917 Geologic Maps
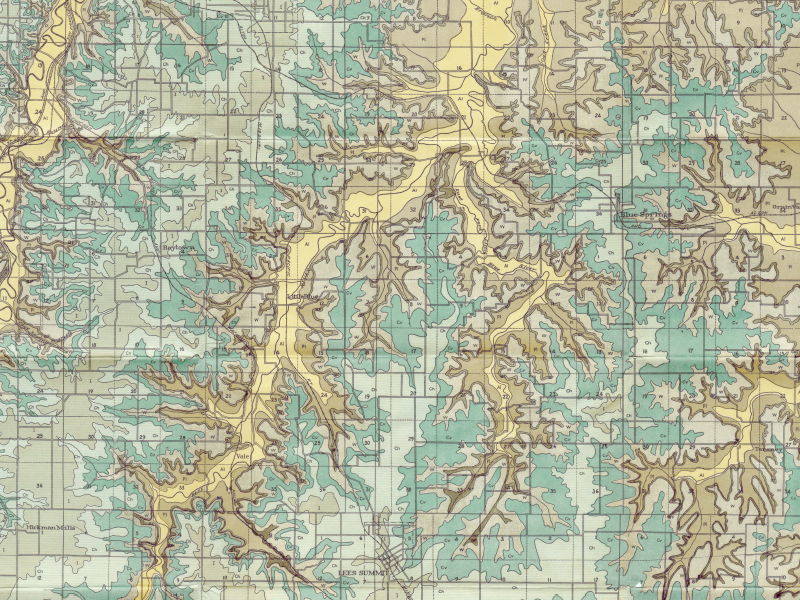 Show-Me Rockhounds member Dan Snow has provided these geologic maps of Kansas City from 1917 which contain topographical, geological, and cross-sectional data. The maps show where to find several different types of rocks common to this area. They are also a great way to see how Jackson County has changed in the last 100 years. The maps are in PDF format and are very high resolution, so please zoom in!
Show-Me Rockhounds member Dan Snow has provided these geologic maps of Kansas City from 1917 which contain topographical, geological, and cross-sectional data. The maps show where to find several different types of rocks common to this area. They are also a great way to see how Jackson County has changed in the last 100 years. The maps are in PDF format and are very high resolution, so please zoom in!
Cross Sections (21 MB)
Jackson County (55 MB)
Kansas City (53 MB)
Fluorite and Calcite on Sphalerite

Photo by Anton Watzl
Here is some fluorite (purple) and calcite (yellow) on sphalerite (silver). No further comments, I just thought this was pretty.
