
Rocks
Fairburn Agate
Fairburn Agate from South Dakota, photographed under white light, then under longwave UV light.
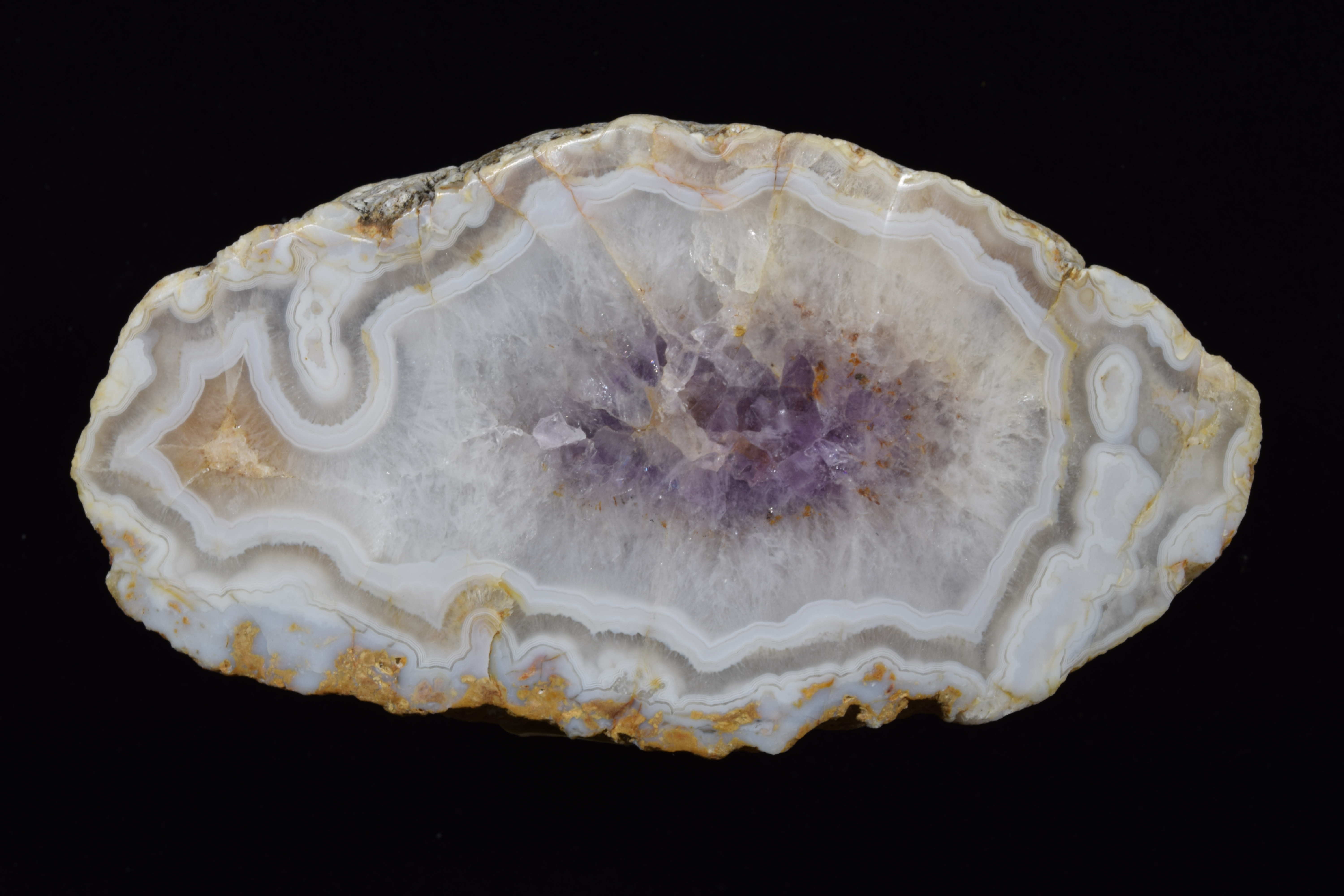
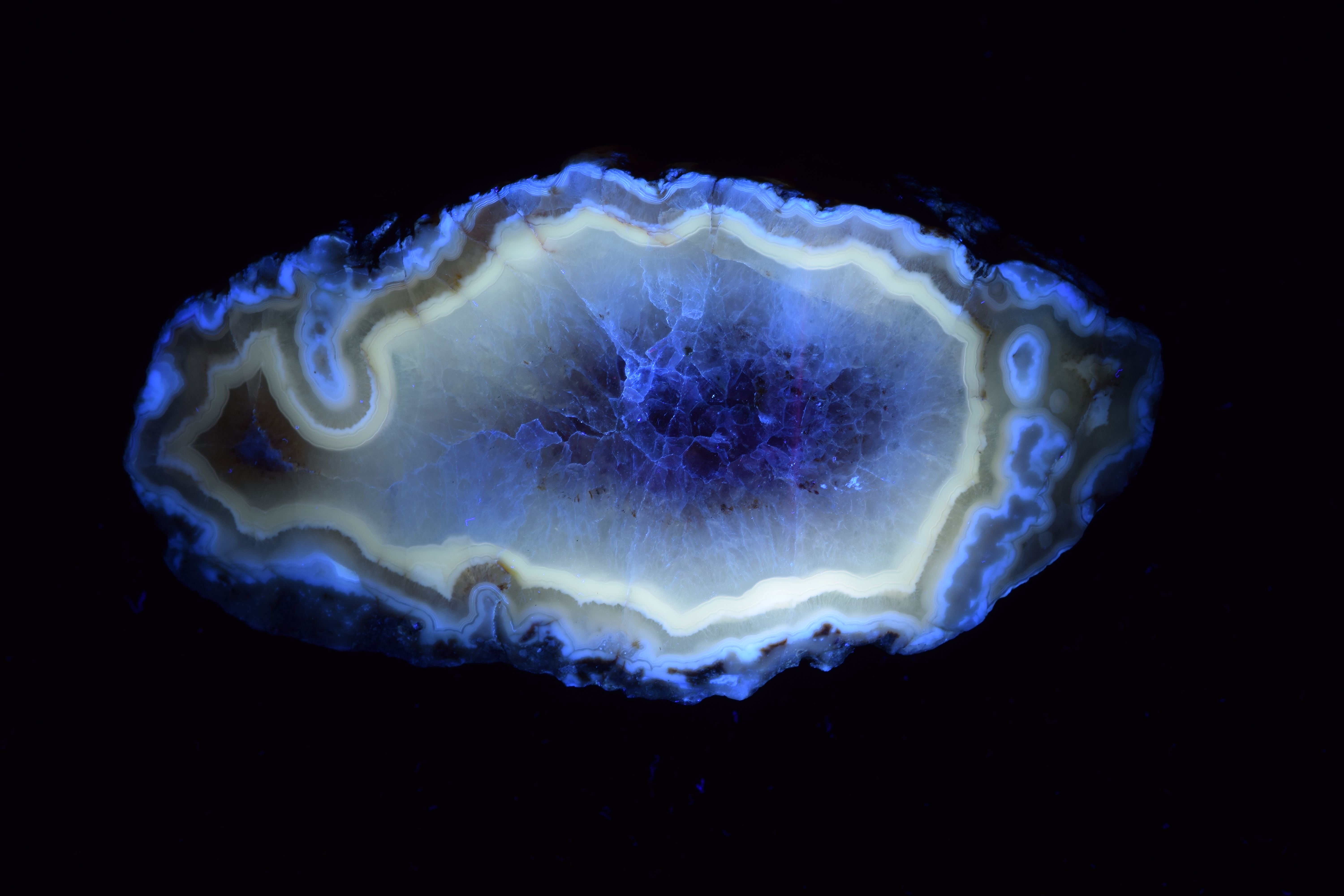
Photos by Dan Snow
This will be on display at the 59th Gem & Mineral Show at the KCI Expo Center, 2020
Chemistry Rocks Webinar

The American Chemical Society is hosting a live webinar called “Chemistry Rocks! – Exploring the Chemistry of Rocks and Minerals” on Tue, Oct 24, 2017 from 6:00 PM – 7:00 PM CDT. We are surrounded by rocks and minerals everywhere…in the ground we walk on, the places we work and live, and even in the food we eat. How are chemists experimenting with these fundamental materials to help the world and make our lives better? Ask questions live to the experts regarding the amazing work that is being done in rock and mineral science.
To see the webinar, sign up at GoToWebinar and fill out the form. They will send you an email to confirm. Then, on Tuesday at 6PM Central Time, follow the link in the email, make sure you have your computer’s sound turned on, and enjoy!
Chemistry in Mining
During Earth Science WeekTM, we went to a lecture by Dr. Innocent Pumure from UCM called “Sonochemical Extraction of Arsenic and Selenium from Pulverized Rocks Associated with Mountaintop Removal Valley Fill (MTR/VF) Method of Coal Mining”.
You may be wondering, what is Mountaintop Removal Valley Fill Mining? First, the excavation company blows up (or strips) the top part of the mountain to remove vegetation and expose the coal seams. The coal seams are then mined through the open cast/strip method, and the extra rock and soil is dumped in nearby valleys called valley fills. It is cheaper and easier to do than regular mining, where they dig a vertical shaft down and do everything through the tunnel, but it blasts the mountain apart and looks ugly. Since 30% of electricity in the USA comes from coal, valley fill mining is still pretty popular.
In 2002, the EPA found too much selenium downstream of a certain mine in West Virginia (we’re not going to say which one). It was over 5 ng/mL, which was the limit back then.[*] 7 years later, there was still an active mine there and the water still had too much selenium. Even worse, the surrounding sediment had 10.7 mg/kg selenium. This could cause problems for the environment later. Due to bioaccumulation, you could say once it’s in there, it’s really in there.
So now we get to the topic of Dr. Pumure’s talk, in which he and his colleagues discovered a way to quickly find out how much selenium and arsenic were in the ground around this mine in West Virginia. When you do a chemical analysis, you usually have to break down the samples in order to measure what is in them. One method to do this would be to take some core samples and do an acid extraction, but that takes a long time and uses a lot of reagents. Sonochemical extraction uses ultrasound energy to accelerate the leaching process that would naturally happen as rocks become weathered. Since it is ultrasound, it does not directly touch the sample, is minimally invasive, and does not need any reagents except water.
Next, he explained the methodology, which means a description of exactly how they did it in the lab: the size of the extraction cells, how much water and power were used (200W/cm3), how long the samples were sonicated, and all the other pertinent information for chemists. Pumure actually spent quite a lot of time finding out the optimal sonicating time to get the best extraction. It turned out the best times for his sample sizes were 20 minutes for Se and 25 minutes for As. That’s really fast![**] Then, he did a comparison to a chemical sequential extraction to make sure that the sonochemical extraction method was getting everything. To summarize, yes it was. Finally, he did a principal component analysis of core samples from different places all over the mountain using this same technique. They found some really interesting trends and correlations, for example, it appears that there is more arsenic in illite clay than other types of clay.
This research has many useful applications. If you were running a mine, you could take samples more frequently to see if your mine is polluting the surrounding environment, and then you could do something about it before the EPA finds out. The method could probably be used for other analytes, too. For other research needs, you could now quickly analyze large batches of mineral samples to get lots of data that would otherwise be too expensive or time consuming to obtain.
[*]The EPA has since lowered the limit and now it is 3.1 ng/mL.
[**]For comparison, some of my colleagues do chemical extractions that take 2 days.
Mini Field Trip July 2017
After our meeting on July 15, 2017 we decided to go on another mini field trip and look for fossils. There were crinoid stems, composita, and other fossils, as well as lots of blue shale. There was also something red and nobody knew what it was. I don’t have many pictures because it was very hot outside and my phone said something about battery temperature too high.

Composita found in Kansas City. Photo by Stephanie Reed

Blue shale. Photo by Stephanie Reed

David met a friendly bird. Photo by David Reed.

This is a traditional pose for members of rock clubs. Someday we may make a calendar. Photo by Stephanie Reed
Mozarkite Trip to Lincoln
MOZARKITE, MISSOURI’S STATE STONE by Roger K. Pabian
(Editor’s note: This article was written by Roger and printed in The Gemrock in 2002. It is written about a field trip taken by Roger and Bill and Betty White.)
On May 3, I took a trip to Kansas City and then on to Lincoln, Missouri, to examine the in place occurrence of Mozarkite, the Official State Gemstone of Missouri. As part of my ongoing study of cryptocrystalline and amorphous quartz family gemstones, I thought that the Mozarkite mine would be a worthwhile trip.
In Kansas City, I joined up with Bill and Betty White on Friday afternoon. Bill and I spent much of the afternoon at one of the major tool houses there and I purchased quite a few diamond tools and other tools that would be of use for stone and metal work. We also hit one of the retail salvage outlets, a store that carries distressed merchandise, as they often have many tools of considerable value for very low prices.
On Saturday morning about 7:00 A.M. we left Independence for the small town of Lincoln, Missouri. The town is famous for its annual rock swap in September. There we teamed up with Linville Harms, owner of the Mozarkite mine, and then went on to the mine. The attached photos of the Mozarkite and the Mozarkite mines help you get a better idea of what the site is like.

Photos by Roger K. Pabian
Mozarkite is not an accepted mineral name but is simply a trade name that was developed to promote the acceptance of the stone as Missouri’s official State Gem and to generate sales to both lapidary and tourists. The name has found acceptance in some circles but is not an acceptable mineral name in others.
Mozarkite has formed in place in marine sedimentary rocks of Ordovician age — it probably is most common in the Jefferson City Formation. The Jefferson City Formation is comprised mostly of dolomite with silty and cherty stringers running through it. There are very few fossils in dolomized rocks as the addition of magnesium to the calcium carbonate of the limestone usually results in complete re-crystallization of the rock and destruction of any fossils or sedimentary structures therein. We did observe a fragment of a brachiopod shell that escaped destruction. It appeared to be a flat-shelled, long-hinge lined form, probably a strophomenoid, but no other determination could be made of it. Much of the local lore about Mozarkite attributes it to igneous activity but there is no evidence for any in that area of Ordovician or younger rocks.
The Mozarkite appears to be of strictly marine sedimentary origin. Some of the nodules show evidence of an accumulation of siliceous gel or ooze on their outer surfaces.
There appears to be three different facies of Mozarkite. The gemmy kind is a dense, brittle form that shows no crystallinity at 10X magnification. A second kind is what the locals call “sugary” Mozarkite. Some of this is quite colorful and has interesting patterns and enjoys some gem use. The “sugary” kind, However, this does not polish nearly as well as the dense, brittle kind. Then, there are some nodules that appear to be very fine sandy textured.
The three facies or textures of Mozarkite suggest that sorting of particles may have been one of the key factors in the origin of the material. Sorting of particles simply means that as some energy form such as wind or flowing water moved a mixture of unconsolidated particles, the heaviest or largest particles are the first ones to drop out of suspension. You can observe this phenomenon on the gravel bars of a stream or in the bars along beaches, estuaries, or lagoons. The coarsest particles will be on the upstream end of the bar or nearer the bottom of the bar. It may well be that the gem Mozarkite is a quartz argillite, a sedimentary rock made up of quartz particles of clay size, that is, smaller than 1/256th of a millimeter. The gemmy facies could also be derived from silica of organic or volcanic origin. The “sugary” facies is made up of the particles larger than 1/16th but smaller than 1/4 mm.
The source for the silica that makes up Mozarkite is currently not known. It may have been from Precambrian granite rocks that are found to the south and east. Sponge spicules may have been the source of silica; I will not totally disregard them. However, I usually favored volcanic ash as the source of siliva for large bodies of chert or flint in marine sedimentary sequences. If there was any volcanic activity involved with Mozarkite, it was from volcanoes that were far away from the Mozarkite-bearing strata.
Mozarkite is a very interesting gem material that could shed a lot of light on the geologic events and processes that led to its formation. My comments above are only a few ideas about its occurrence. Like many other ideas on his stone, my hypotheses need more documentation before they can either be accepted or rejected. My hypotheses should probably read as follows: “Mozarkite is a quartz argillite of marine sedimentary origin that formed in situ in shallow seas of Ordovician age. The source of the quartz is shield rocks of Precambrian ages that lie to the southeast of the area from which it is not found.”
To prove that, several things need to be done. First, properly oriented (top and north) nodules need to be collected from in place in the mine pits. The nodules should not be examined in the field to avoid “high grading” the material. An outcrops map or diagram would need to be made that shows the places from which each nodule was taken. Similar sampling should be carried out from several different layers in several different parts of the mine. The facies of each nodule would need to be located on the map. Does one zone produce only sandy material whereas another produces only gemmy material? Or do these facies occur at random? Thin sections (30 microns) would have to be made. The nature of the particles (angular or rounded) and any cement between them would need to be noted. Is there a silica cement between the particles or does their angularity hold them together? Then other occurrences, both geographic and stratigraphic, of Mozarkite would have to be noted. The sedimentary structures in the Mozarkite and the host rock would also have to be observed and recorded.
By the time all of this is done, one has done enough work to earn a Master of Science Degree. As you see, there is no easy answer for Mozarkite. Perhaps, as a club, or group of clubs, we might think of funding a student to carry out the above kind of research.

Linville Harms (left) of Sedalia, Missouri, and Bill & Betty White examine the open pit mine. Linville is the mine owner. Photo by Roger K. Pabian
1917 Geologic Maps
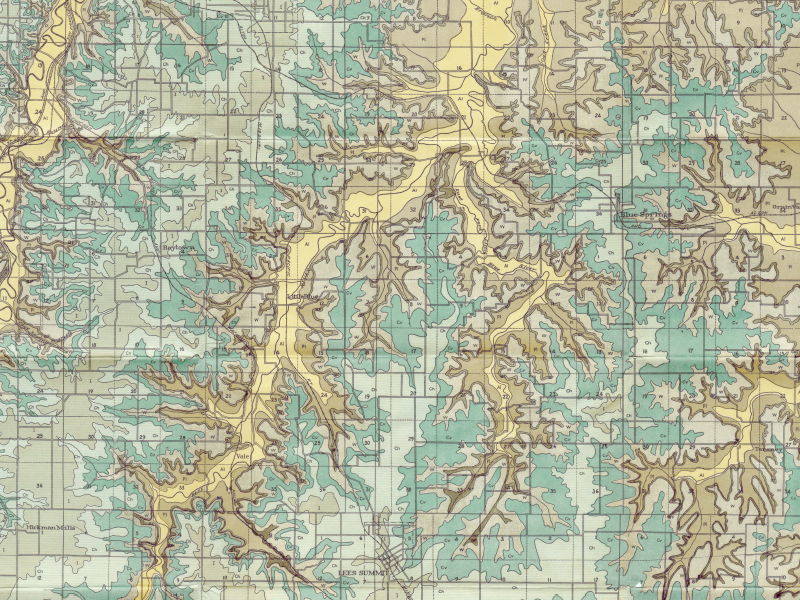 Show-Me Rockhounds member Dan Snow has provided these geologic maps of Kansas City from 1917 which contain topographical, geological, and cross-sectional data. The maps show where to find several different types of rocks common to this area. They are also a great way to see how Jackson County has changed in the last 100 years. The maps are in PDF format and are very high resolution, so please zoom in!
Show-Me Rockhounds member Dan Snow has provided these geologic maps of Kansas City from 1917 which contain topographical, geological, and cross-sectional data. The maps show where to find several different types of rocks common to this area. They are also a great way to see how Jackson County has changed in the last 100 years. The maps are in PDF format and are very high resolution, so please zoom in!
Cross Sections (21 MB)
Jackson County (55 MB)
Kansas City (53 MB)
Hematite Magnetite

Photo by Stephanie Reed
This hematite and magnetite specimen is from Patagonia, Argentina. Bruce got it at the Denver show and gave it to Sharon Penner. It’s about 4 inches long and pretty shiny.
Fairburn Agates
Special guest article from Show-Me Rockhounds club members Dan and Connie Snow

Group picture of Fairburn agates – Dan Snow
Fairburn agates are a form of microcrystalline chalcedony, 100% silicon dioxide with a hardness of 6 ½ to 7 on the Mohs scale. They are also called fortification agates because of their banding. They were formed approximately 300 million years ago in an ancient limestone bed of an inland sea. To hunt Fairburn agates requires looking at every rock and turning many with a rock pick. It is strictly surface hunting no digging, mining, cracking or breaking rocks. The photos shown are exactly the way the agates were found, with no cutting, polishing or tumbling having been done.

Frog Rock – Dan Snow
Fairburn Agates found by Dan and Connie Snow. Collected from the Fairburn Agate beds of South Dakota and the Oglala National Grasslands in Nebraska.
Labradorite
Labradorite has become a popular gemstone because of the unique iridescent play of color that many specimens exhibit. Labradorite is a feldspar mineral of the plagioclase series that is most often found in mafic igneous rocks such as basalt, gabbro and norite. Some specimens of labradorite exhibit a Schiller effect, which is a strong play of iridescent blue, green, red, orange, and yellow colors as shown in the photographs above. The Schiller effect is also seen in fire agate and mother of pearl. Labradorite is so well known for these spectacular displays of color that the phenomenon is known as “labradorescence.” Specimens with the highest quality labradorescence are often selected for use as gemstones. Labradorescence is not a display of colors reflected from the surface of a specimen. Instead, light enters the stone, strikes a twinning surface within the stone, and reflects from it. The color seen by the observer is the color of light reflected from that twinning surface. Different twinning surfaces within the stone reflect different colors of light. Light reflecting from different twinning surfaces in various parts of the stone can give the stone a multi-colored appearance.







